
Why this matters
Automatic Continuous Online Monitoring of Polymerizations (ACOMP) was used to monitor the free radical polymerization of polyvinyl pyrrolidone (PVP). The physical properties of a polymer such as brittleness, conductivity or solubility are determined by the molecular weight distribution (MWD) and its various averages, especially weight-average molecular weight (Mw). The real-time monitoring of polymerization reactions is important in many industrial and research applications from new product development and pilot reactions scale-up to the optimization and quality control of existing processes.
Background
ACOMP was used to monitor the free radical polymerization of polyvinyl pyrrolidone using UV Absorption (UV), capillary viscometry and multi-angle light scattering (MALS). This technique is suitable for many processes, including solution, bulk, emulsion and inverse emulsion polymerizations, as well as post-processing steps such as hydrolysis and quaternization.
Polyvinyl pyrrolidone (PVP) is a versatile water-soluble polymer that is used in many applications, including filtration, pharmaceutical tablet coatings, binder in personal care products, dishwasher soap packaging, inks and coatings. Increased demand for this versatile polymer calls for the fusion of analytical, sensor and process data, yielding correlations that can optimize product quality and increase plant throughput, while simultaneously reducing energy and feedstock consumption.
This technical note describes how ACOMP is used to monitor the polymerization of vinyl pyrrolidone under small changes in initiator concentrations.
Methodology
To demonstrate the applicability of ACOMP to the polymerization of polyvinyl pyrrolidone, two batch reactions using 3% vinyl pyrrolidone (VP) were run at 1.5% and 2.5% initiator concentration to monitor the effects of a small change in initiator concentration on reaction kinetics, weight average molecular weight and intrinsic viscosity. The reactions were conducted at a constant temperature of 45°C.
A UV Absorption detector was used to measure the concentration of the vinyl pyrrolidone monomer at 260 nm. A Fluence Analytics proprietary viscometer was used to measure the intrinsic viscosity, and a proprietary 5-angle light scattering (MALS) detector was used to measure weight average molecular weight. The ACOMP system interfaced with the reactor through a recirculation loop, ensuring a representative sample of the reactor contents was analyzed. The recirculation loop was continuously sampled at 0.5 ml/min, and the extracted sample was diluted with de-ionized water to the appropriate detector concentrations. The sample was diluted 12 times to the viscometer and 80 times to the UV and MALS detectors.
Generally, ACOMP dilutions range from 10X to 1,000X dilution depending on the reactor concentration and the properties of the polymer.
Figure 1 shows the typical ACOMP Raw Signals for the UV, viscometer and light scattering of the polymerization of vinyl pyrrolidone. The raw data is generated in real time and can be viewed both through the ACOMP HMI (Human-Machine Interface) and on a linked PC. In an industrial environment, this information is also available instantaneously to the process and quality control engineers through the plant’s distributed control system (DCS). A baseline, indicative of the reactor conditions prior to polymerization, is collected for all the detectors. Once the reaction is initiated, UV decreases with monomer consumption as displayed in blue, and viscometer and light scattering measurements increase throughout the polymerization as depicted in red and green, respectively.
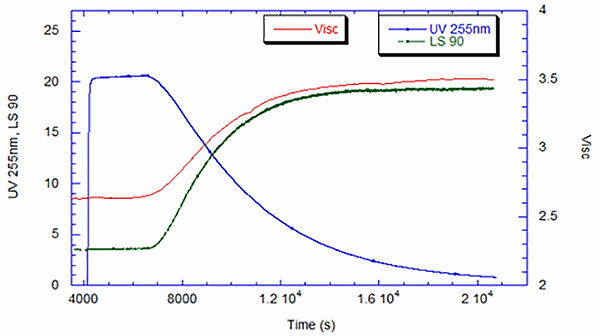
Polyvinyl Pyrrolidone Reaction Kinetics
Figure 2 exhibits the polymer concentration as measured by the UV for both reactions. The reaction was initiated after the monomer baseline was determined stable, which occurred around 3000 seconds. As expected, the reaction with 2.5% initiator shows much faster conversion to polymer and closely adheres to first-order reaction kinetics. At 1.5% initiator, the reaction begins more slowly, perhaps due to impurities that interfere with the initiator and are more prevalent at the lower concentration. This type of insight is very valuable for process engineers operating a batch process of this type and for R&D chemists working to scale-up new recipes.
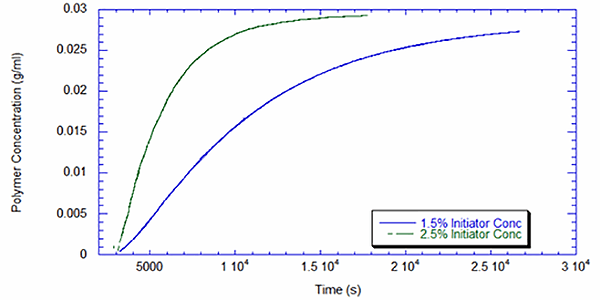
Intrinsic Viscosity and Molecular Weight
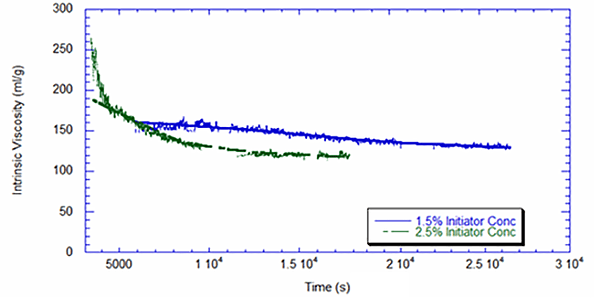
Figure 3 displays the intrinsic viscosity as measured by the viscometer. The higher initiator concentration leads to a 10% decrease in intrinsic viscosity.
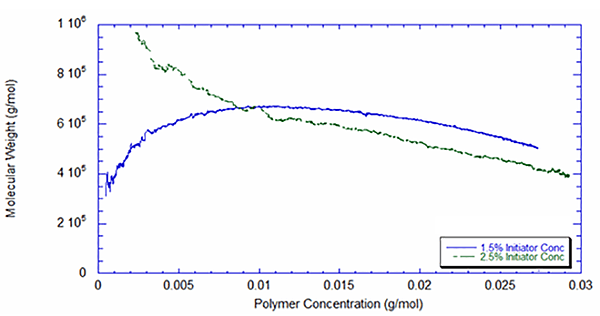
Figures 4 and 5 present the weight average molecular weight as measured by the light scattering detector, combined with the UV detector. The difference in behavior for the two reactions is noteworthy. With 2.5% initiator, the molecular weight roughly follows what would be expected in a batch reaction with first-order kinetics, but in the reaction with 1.5% initiator, the molecular weight starts low and peaks at around two hours. Once again this suggests the likelihood that the reaction at a lower initiator level is inhibited by impurities at the outset. It is also interesting that the higher initiator concentration leads to a decrease in molecular weight – roughly 23% lower at the end of the reaction, as shown in Table 1.
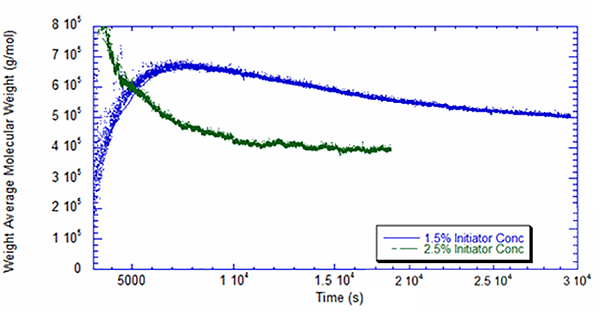

Conclusion
ACOMP can successfully monitor the polymerization of polyvinyl pyrrolidone under varying initiator concentrations. In industrial applications, operators can identify and take corrective action much sooner in the event of an undesirable process condition such as a change in initiator effectiveness due to impurities. ACOMP is also useful for the real-time monitoring of other polymer properties like radius of gyration, hydrodynamic value, branching and degree of crosslinking, which greatly influence final product performance. Additional technical notes demonstrating ACOMP’s value in monitoring polymers used in rubber, paint, adhesive and plastic applications is available here.
References
1. Reed, W.F. “Automated Continuous Online Monitoring of Polymerization Reactions (ACOMP) and Related Techniques.” Encyclopedia of Analytical Chemistry, 2013, 1-40, DOI: 10.1002/9780470027318.a9288.
2. Florenzano, F. H., Strelitzki, R., & Reed, W. F. (1998). Absolute, On-Line Monitoring of Molar Mass during Polymerization Reactions. Macromolecules, 31(21), 7226-7238. doi:10.1021/ma980876e

